Structural Basis for Adenylate Kinase Activity in ABC ATPases
13-Aug-2010
Journal of Molecular Biology, 2010, 401, 2, 265 - 73 published on 13.08.2010
Journal of Molecular Biology, online article
Journal of Molecular Biology, online article
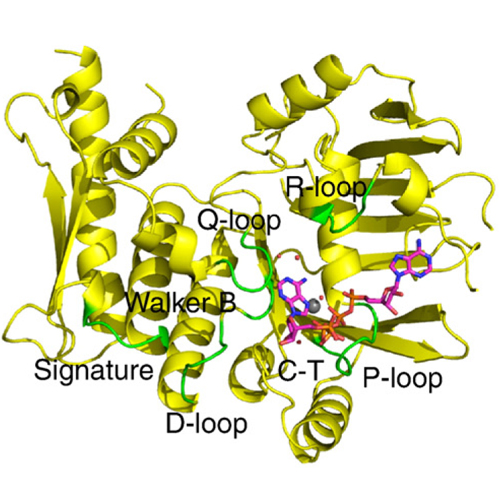
ATP-binding cassette (ABC) enzymes are involved in diverse biological processes ranging from transmembrane transport to chromosome cohesion and DNA repair. They typically use ATP hydrolysis to conduct energy-dependent biological reactions. However, the cystic fibrosis transmembrane conductance regulator and the DNA repair protein Rad50 can also catalyze the adenylate kinase reaction (ATP + AMP ↔ 2ADP). To clarify and provide a mechanistic basis for the adenylate kinase activity of ABC enzymes, we report the crystal structure of the nucleotide-binding domain of the Pyrococcus furiosus structural maintenance of chromosome protein (pfSMCnbd) in complex with the adenylate kinase inhibitor P1,P5-di(adenosine-5′)pentaphosphate. We show that pfSMCnbd possesses reverse adenylate kinase activity. Our results suggest that in adenylate kinase reactions, ATP binds to its canonical binding site while AMP binds to the Q-loop glutamine and a hydration water of the Mg2+ ion. Furthermore, mutational analysis indicates that adenylate kinase reaction occurs in the engaged pfSMCnbd dimer and requires the Signature motif for phosphate transfer. Our results explain how ATP hydrolysis and adenylate kinase reactions can be catalyzed by the same functional motifs within the structural framework of ABC enzymes. Thus, adenylate kinase activity is likely to be a latent activity in many ABC enzymes.