Insights into the Mode of Action of Novel Fluoroketolides, Potent Inhibitors of Bacterial Protein Synthesis
04-Nov-2013
American Society for Microbiology, 2013, doi: 10.1128/AAC.01994-13, vol. 58 no. 1 472-480 published on 04.11.2013
American Society for Microbiology, online article
American Society for Microbiology, online article
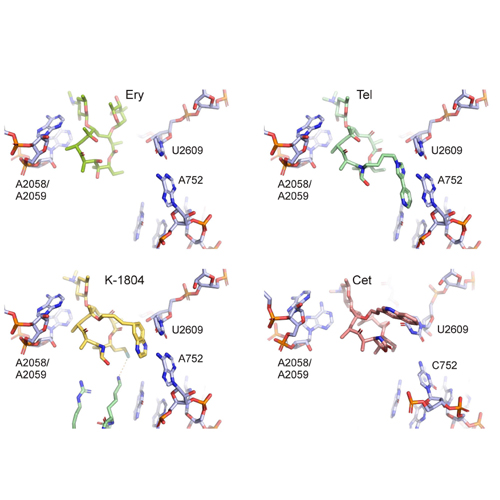
Ketolides, the third generation of expanded-spectrum macrolides, have in the last years become a successful weapon in the endless war against macrolide-resistant pathogens. Ketolides are semisynthetic derivatives of the naturally produced macrolide erythromycin, displaying not only improved activity against some erythromycin-resistant strains but also increased bactericidal activity as well as inhibitory effects at lower drug concentrations. In this study, we present a series of novel ketolides carrying alkyl-aryl side chains at the C-6 position of the lactone ring and, additionally, one or two fluorine atoms attached either directly to the lactone ring at the C-2 position or indirectly via the C-13 position. According to our genetic and biochemical studies, these novel ketolides occupy the known macrolide binding site at the entrance of the ribosomal tunnel and exhibit lower MIC values against wild-type or mutant strains than erythromycin. In most cases, the ketolides display activities comparable to or better than the clinically used ketolide telithromycin. Chemical protection experiments using Escherichia coli ribosomes bearing U2609C or U754A mutations in 23S rRNA suggest that the alkyl-aryl side chain establishes an interaction with the U2609-A752 base pair, analogous to that observed with telithromycin but unlike the interactions formed by cethromycin. These findings reemphasize the versatility of the alkyl-aryl side chains with respect to species specificity, which will be important for future design of improved antimicrobial agents.