Immunomodulatory drugs disrupt the cereblon–CD147–MCT1 axis to exert antitumor activity and teratogenicity
13-Jun-2016
Nature Medicine, 22, 735–743, doi:10.1038/nm.4128
Nature Medicine, online article
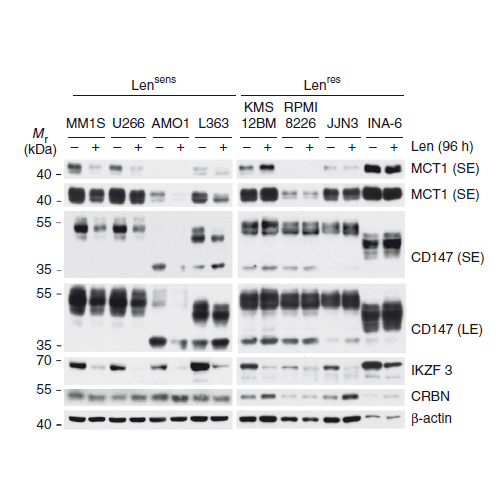
Immunomodulatory drugs (IMiDs), such as thalidomide and its derivatives lenalidomide and pomalidomide, are key treatment modalities for hematologic malignancies, particularly multiple myeloma (MM) and del(5q) myelodysplastic syndrome (MDS). Cereblon (CRBN), a substrate receptor of the CRL4 ubiquitin ligase complex, is the primary target by which IMiDs mediate anticancer and teratogenic effects. Here we identify a ubiquitin-independent physiological chaperone-like function of CRBN that promotes maturation of the basigin (BSG; also known as CD147) and solute carrier family 16 member 1 (SLC16A1; also known as MCT1) proteins. This process allows for the formation and activation of the CD147–MCT1 transmembrane complex, which promotes various biological functions, including angiogenesis, proliferation, invasion and lactate export. We found that IMiDs outcompete CRBN for binding to CD147 and MCT1, leading to destabilization of the CD147–MCT1 complex. Accordingly, IMiD-sensitive MM cells lose CD147 and MCT1 expression after being exposed to IMiDs, whereas IMiD-resistant cells retain their expression. Furthermore, del(5q) MDS cells have elevated CD147 expression, which is attenuated after IMiD treatment. Finally, we show that BSG (CD147) knockdown phenocopies the teratogenic effects of thalidomide exposure in zebrafish. These findings provide a common mechanistic framework to explain both the teratogenic and pleiotropic antitumor effects of IMiDs.